Tirzepatide is a new GIP (gastric inhibitory peptide) and GLP-1 (glucagon-like peptide-1) dual receptor agonist developed and successfully marketed by Eli Lilly and Company for the targeted treatment of diabetes. , non-alcoholic steatohepatitis (NASH) and chronic weight management, with a half-life of 116.7 hours. After being approved to treat diabetes in 2022, it was approved for weight loss in the United States in November 2023. One large clinical trial reported that people using the drug lost
To achieve dual activity, tilpotide not only fused amino acid residues from GLP-1 and GIP, but also used some unique amino acid residues. Its peptide construction is mainly derived from GLP-1, GIP, exenatide and semaglutide, with a few residues being unique.
In this issue, the editor will introduce to you the research on the synthesis process of Eli Lilly's blockbuster drug that has become popular recently: tirzepatide. Currently, many methods have been used to manufacture synthetic peptides. The selection of appropriate synthesis methods is usually based on molecular characteristics. , such as the presence of non-coded amino acids, the presence of non-peptide side chains and the number of amino acids present.
Biosynthesis: Recombinant techniques typically used for large molecules such as monoclonal antibodies can also be applied to smaller peptides. In this approach, external DNA is transplanted into E. coli or mammalian (Chinese Hamster Ovary, CHO) cell lines and the peptides are synthesized by the cells. Development times are generally longer than typical synthetic methods, but the method has been used successfully with semisynthetic peptides such as semaglutide, insulin degludec, and liraglutide. Two disadvantages of this method are: (1) it is difficult to introduce amino acids that are not encoded by DNA sequences; (2) the introduction of non-peptide side chains requires post-recombination synthetic modification.
Chemical synthesis:
Solid-phase peptide synthesis (SPPS). It has been shown to be effective against a number of medium molecules (e.g., lixisenatide and exenatide), including the use of non-coded amino acids in the synthesis process. Unfortunately, for larger peptides (usually >30 amino acids), SPPS often suffers from low process yields and low purity, and due to the need to perform a large number of consecutive unit operations without errors (for >30 nucleic acid peptides, with an estimated failure rate of 20%) and therefore pose significant production risks.
Liquid phase peptide synthesis (LPPS). The method is similar to SPPS, except that the growing peptide is not bound to the resin and the C-terminus of the peptide is a non-reactive amide or protected ester. This method works well for the synthesis of smaller peptides (≤10 amino acids), but yields and purity decrease rapidly as the peptides become larger.
SPPS/LPPS hybrid method. This approach creates highly pure shorter peptide fragments from SPPS as well as well-characterized precipitated intermediates, avoiding many of the manufacturing risks and purity issues inherent in longer SPPS constructs. The protected fragments are then coupled in the liquid phase.
The SPPS/LPPS hybrid method combines the advantages and disadvantages of SPPS and LPPS and is a good choice. To implement the hybrid method process, the first step is to select the appropriate fragments that will be synthesized by LPPS. Key considerations for fragment selection are peptide length, total number of fragments, identity of the amino acids at the break point, and side chains. The size and number of fragments are important because many smaller fragments, while highly pure, result in a more tedious manufacturing process with more LPPS steps. Fewer large fragments, while having less manufacturing burden, is inherently more difficult to obtain high purity with higher manufacturing risk. Balancing these considerations, Lilly researchers selected four fragments for use in the synthesis of tilpotide. For breakpoints, a critical aspect is the epimerization of the C-terminal amino acids in each fragment, since the amide backbone makes the α-steric center more susceptible to epimerization compared to the Fmoc-protected amino acids used in the SPPS construction. Heterogeneous. After exploring various fragments, four fragments were selected (Figures 2−5 below). All four fragments are synthesized via SPPS as easily isolated solids and can be obtained in extremely high purity (97.5−99.5% purity).
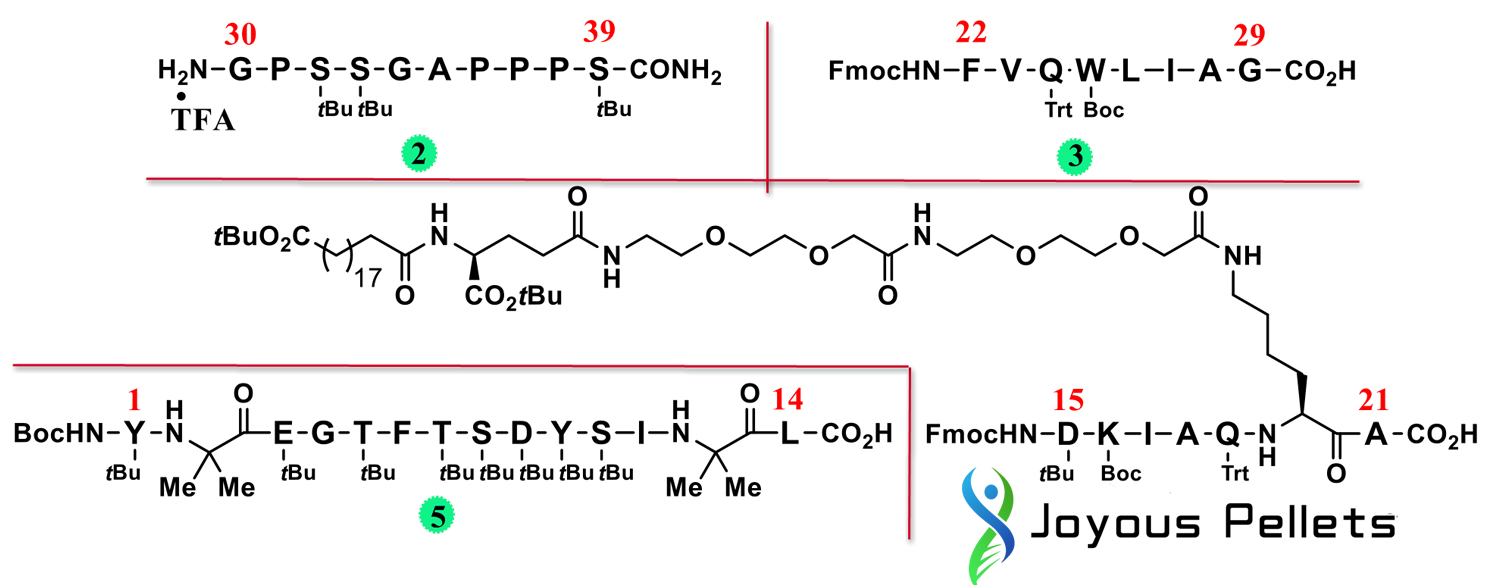
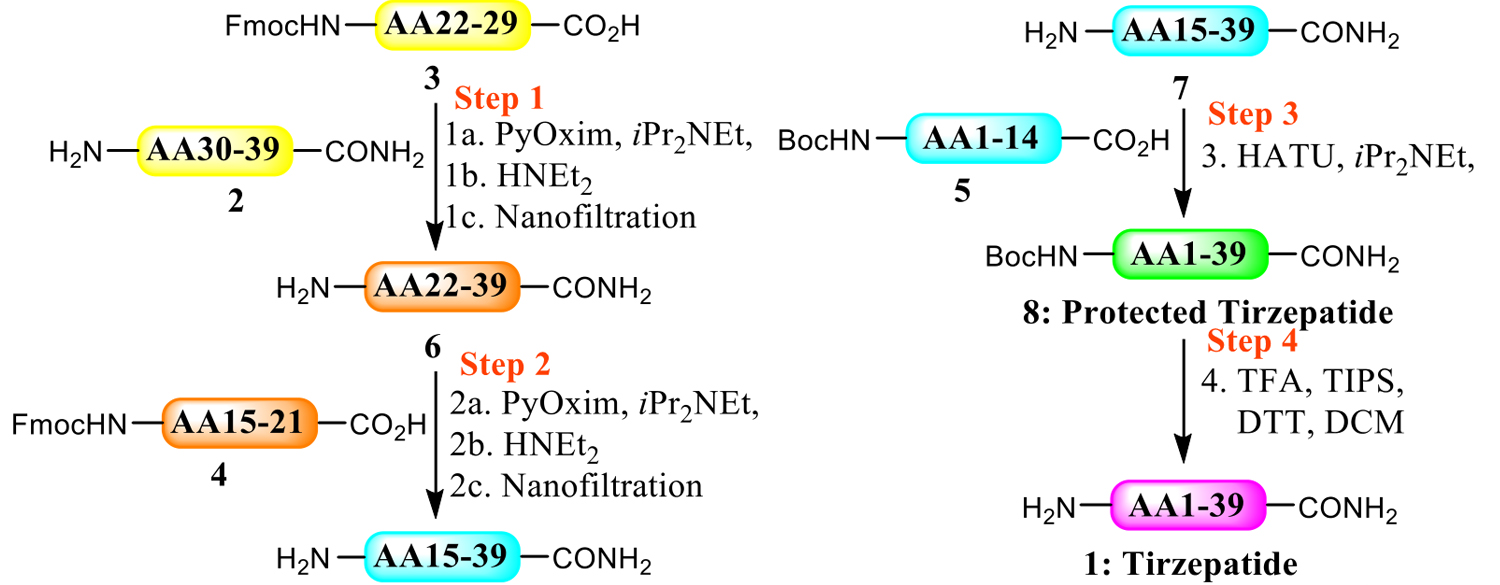
Next, fragments 2-5 were coupled together through four steps of LPPS.
Step 1: Connect the first two fragments 2 and 3, representing amino acids 30−39 and 22−29 respectively. Separate solutions of 2 and 3 in dimethyl sulfoxide (DMSO) and acetonitrile (ACN), PyOxim in ACN, and pure diisopropylethylamine were prepared in advance and fed into the plug flow reactor (PFR) 27 Before flowing together, in-line mixing is performed to ensure uniformity;
Step 2: The obtained amino acids 22-39 (compound 6) are then coupled with fragment 4 to obtain amino acids 15-39 (compound 7);
Step 3: The obtained amino acids 15−39 (compound 7) are coupled with fragment 5 to obtain Boc-protected tilpotide;
Step 4: Deprotect to obtain tilpotide.
Nanofiltration is a membrane-based technology in which dissolved substances in a solution can be separated based on molecular weight and hydrophobicity. The crude reaction solution passes through the membrane to remove small molecular weight impurities (such as dibenzofulene (DBF), diethylamine (DEA) and PyOxim-related by-products (PyO)) while retaining the desired peptides. The choice of pore size is very important. , and need to balance low throughput (if the pore size is too small) with peptide loss (if the pore size is too large). Based on such a technology, many operations can be simplified in mass production. Interested children can read the original link on the lower left.
Since tilpotide is purely chemically synthesized, there are many corresponding companies for tilpotide API, and competition is fierce. Currently, tilpotide API is produced domestically by Hequan Pharmaceuticals and shipped abroad. Prepare preparations. Due to the maturity of chemical synthesis and the large number of CRO manufacturers, Joyous believes that tilpotide will have greater production capacity compared to semaglutide.

